
It occurs naturally only in compounds with other elements such as strontianite. 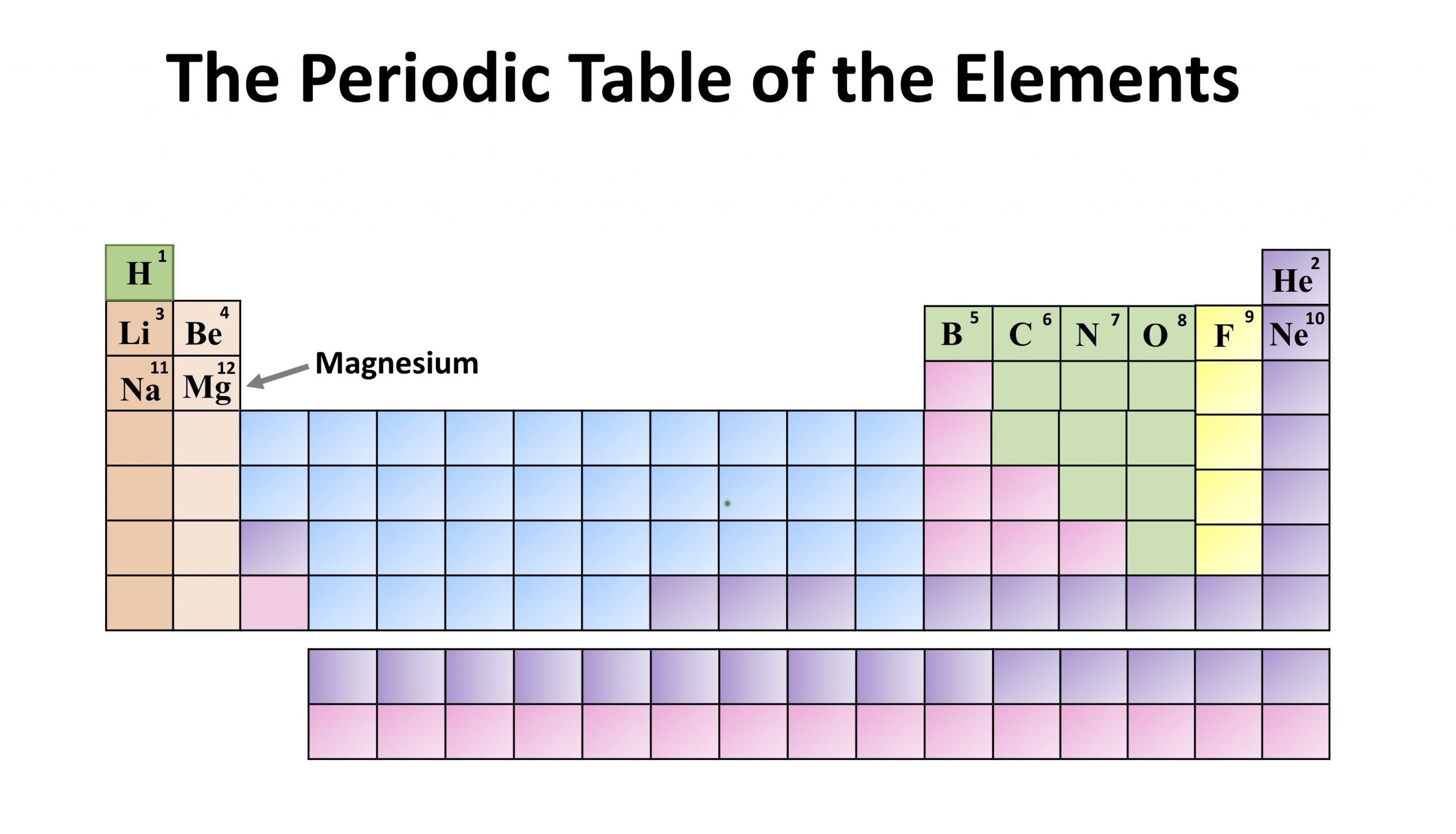
Chemistry of Strontium (Z=38) Strontium is a group 2 element that does not occur as a free element due to its extreme reactivity with oxygen and water.All living organisms (in fact, even dead ones) have and need calcium for survival. Calcium is the fifth most abundant element by mass (3.4%) in both the Earth's crust and in seawater. It is a group 2 metal, also known as an alkaline-earth metal, and no populated d-orbital electrons. Chemistry of Calcium (Z=20) Calcium is the 20th element in the periodic table.Fine particles of magnesium can also catch on fire when exposed to air. Like aluminum, it forms a thin layer around itself to help prevent itself from rusting when exposed to air. Magnesium is light, silvery-white, and tough. Chemistry of Magnesium (Z=12) Magnesium is a group two element and is the eighth most common element in the earth's crust.Some Atypical Properties of Beryllium Compounds.It is not abundant in the environment and occurs mainly in the mineral beryl with aluminum and silicon. The element is a high-melting, silver-white metal which is the first member of the alkaline earth metals. Chemistry of Beryllium (Z=4) The name Beryllium comes from the Greek beryllos which is the name for the gemstone beryl.Sections below cover the trends in atomic radius, first ionization energy, electronegativity, and physical properties. Group 2: Physical Properties of Alkali Earth Metals This page explores the trends in some atomic and physical properties of the Group 2 elements: beryllium, magnesium, calcium, strontium and barium.The Thermal Stability of the Nitrates and Carbonates.The Solubility of the Hydroxides, Sulfates and Carbonates.Reactions of Group 2 Elements with Water.Reactions of Group 2 Elements with Oxygen.Reactions of Group 2 Elements with Acids.Includes trends in atomic and physical properties, trends in reactivity, the solubility patterns in the hydroxides and sulfates, trends in the thermal decomposition of the nitrates and carbonates, and some of the atypical properties of beryllium. Group 2: Chemical Properties of Alkali Earth Metals Covers the elements beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr) and barium (Ba).This fact has key implications for the building up of the periodic table of elements.\) The ordering of the electrons in the ground state of multielectron atoms, starts with the lowest energy state (ground state) and moves progressively from there up the energy scale until each of the atom’s electrons has been assigned a unique set of quantum numbers. It is the Pauli exclusion principle that requires the electrons in an atom to occupy different energy levels instead of them all condensing in the ground state. In the periodic table, the elements are listed in order of increasing atomic number Z. The number of electrons in each element’s electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. The configuration of these electrons follows from the principles of quantum mechanics. The chemical properties of the atom are determined by the number of protons, in fact, by number and arrangement of electrons. See also: Atomic Number – Does it conserve in a nuclear reaction? Atomic Number and Chemical PropertiesĮvery solid, liquid, gas, and plasma is composed of neutral or ionized atoms. It is the electrons that are responsible for the chemical bavavior of atoms, and which identify the various chemical elements. In a neutral atom there are as many electrons as protons moving about nucleus. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The nucleus is composed of protons and neutrons. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
